Start your patients on KISQALI with confidence
.
SIMPLIFIED TREATMENT INITIATION

A few standard assessments help to ensure your patients start right away
- Complete blood count (CBC)1
- Liver function tests (LFTs)1,*
- Electrolyte levels1,†
- Electrocardiogram (ECG)1,‡

Straightforward ECG testing
- The 2 required ECGs are both completed within the first 2 weeks of treatment1
- If you are unable to perform ECGs in office, speak with your Novartis Oncology Specialist about a simple solution for fast, easy, and accurate ECG testing
*Monitor LFTs prior to the initiation of treatment, every 2 weeks for the first 2 cycles, at the beginning of each of the subsequent 4 cycles, and as clinically indicated. For LFTs, if grade ≥2 abnormalities are noted, more frequent monitoring is recommended.1
†Monitor serum electrolytes prior to the initiation of treatment, at the beginning of the first 6 cycles, and as clinically indicated. Correct any electrolyte abnormalities before initiating treatment.1
‡KISQALI should only be initiated in patients with QTcF <450 ms.1
Additional monitoring may be required as clinically indicated.
§There were no reported cases of torsades de pointes. KISQALI has been shown to prolong the QT interval in a concentration-dependent manner. In MONALEESA-2, there was 1 (0.3%) sudden death in a patient with grade 3 hypokalemia and grade 2 QT prolongation.1
DOSING & ADJUSTMENTS
For your patients with HR+/HER2- mBC,
Start with KISQALI 600 mg—the starting dose with proven outcomes
KISQALI single-strength tablets make dose reduction simple and convenient1
KISQALI is given as 600 mg (3 x 200-mg tablets) orally, once daily (3 weeks on, 1 week off) with either1:
An AI once daily (continuously); in men and premenopausal women, an LHRH agonist should also be administered according to current clinical practice guidelines; or
Fulvestrant 500 mg intramuscularly on Days 1, 15, and 29, and once monthly thereafter; in men and premenopausal women, an LHRH agonist should also be administered according to current clinical practice guidelines
Patients should continue treatment until disease progression or unacceptable toxicity1
Dose adjustments for adverse reactions should be made in a stepwise order by reducing the number of tablets taken. Dose modification of KISQALI is recommended based on individual safety and tolerability. If dose reduction below 200 mg/day is required, discontinue treatment.
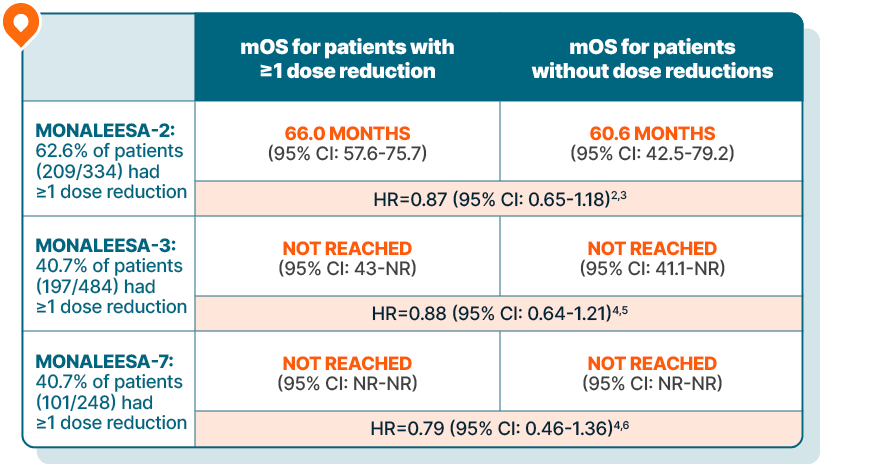
KISQALI maintained overall survival in patients requiring dose reductions across 3 phase III trials
Results are based on a post hoc analysis; efficacy in the placebo comparator arms was not assessed and should be interpreted with caution.

In MONALEESA-2, managing adverse reactions with dose reductions helped patients stay on therapy an average of 6.5 months longer than those without dose reductions.3
MONALEESA-2 was a randomized, double-blind, placebo-controlled, phase III study of KISQALI + letrozole (n=334) vs placebo + letrozole (n=334) in postmenopausal patients with HR+/HER2- mBC who received no prior therapy for advanced disease. OS was a secondary end point; PFS was the primary end point. At a median follow-up of 80 months, mOS was 63.9 months with KISQALI + letrozole (95% CI: 52.4-71.0) vs 51.4 months with placebo + letrozole (95% CI: 47.2-59.7); HR=0.765 (95% CI: 0.628-0.932); P=0.004.1,7,8
MONALEESA-3 was a randomized, double-blind, placebo-controlled, phase III study of KISQALI + fulvestrant (n=484) vs placebo + fulvestrant (n=242) in postmenopausal patients with HR+/HER2- mBC who received no or only 1 line of prior ET for advanced disease. OS was a secondary end point; PFS was the primary end point. In an exploratory analysis of a 1L subgroup of patients receiving KISQALI + fulvestrant (n=237) or placebo + fulvestrant (n=128), at a median follow-up of 71 months mOS was 67.6 months with KISQALI + fulvestrant (95% CI: 59.6-NR) vs 51.8 months with placebo + fulvestrant (95% CI: 40.4-61.2); HR=0.673 (95% CI: 0.504-0.899). At a median follow-up of 39 months, statistical significance was established for overall survival in the ITT population; HR=0.724 (95% CI: 0.568-0.924); P=0.00455. Results from the 71-month analysis were not prespecified and were observational in nature; as such, there was no prespecified statistical procedure controlling for type 1 error.1,9-11
MONALEESA-7 was a randomized, double-blind, placebo-controlled, phase III study of KISQALI + ET (NSAI or tamoxifen) + goserelin (n=335) vs placebo + ET (NSAI or tamoxifen) + goserelin (n=337) (ITT) in premenopausal patients with HR+/HER2- mBC who received no prior ET for advanced disease. KISQALI is not indicated for concomitant use with tamoxifen. Efficacy results are from a prespecified subgroup analysis of 495 patients who received KISQALI (n=248) or placebo (n=247) with an NSAI + goserelin and were not powered to show statistical significance. OS was a secondary end point; PFS was the primary end point. At a median follow-up of 54 months (exploratory analysis), mOS was 58.7 months with KISQALI + NSAI + goserelin (95% CI: 48.5-NR) vs 47.7 months with placebo + NSAI + goserelin (95% CI: 41.2-55.4); HR=0.798 (95% CI: 0.615-1.035). At a median follow-up of 35 months, statistical significance was established for overall survival in the ITT population; HR=0.71 (95% CI: 0.54-0.95); P=0.00973. Results from the 54-month analysis were not prespecified and were observational in nature; as such, there was no prespecified statistical procedure controlling for type 1 error.1,12-15
NOVARTIS PATIENT SUPPORT
A dedicated team for you and your patients
Novartis Patient Support™ is a comprehensive program that is designed to help your eligible patients start, stay, and save on KISQALI.
Your practice and patients will have access to a Novartis Patient Support team committed to providing the support you need, including:
Dedicated assistance with access and reimbursement
Assistance with relevant savings options for your eligible patients
Personalized support for your patients on therapy
Single points of contact for you and your patients